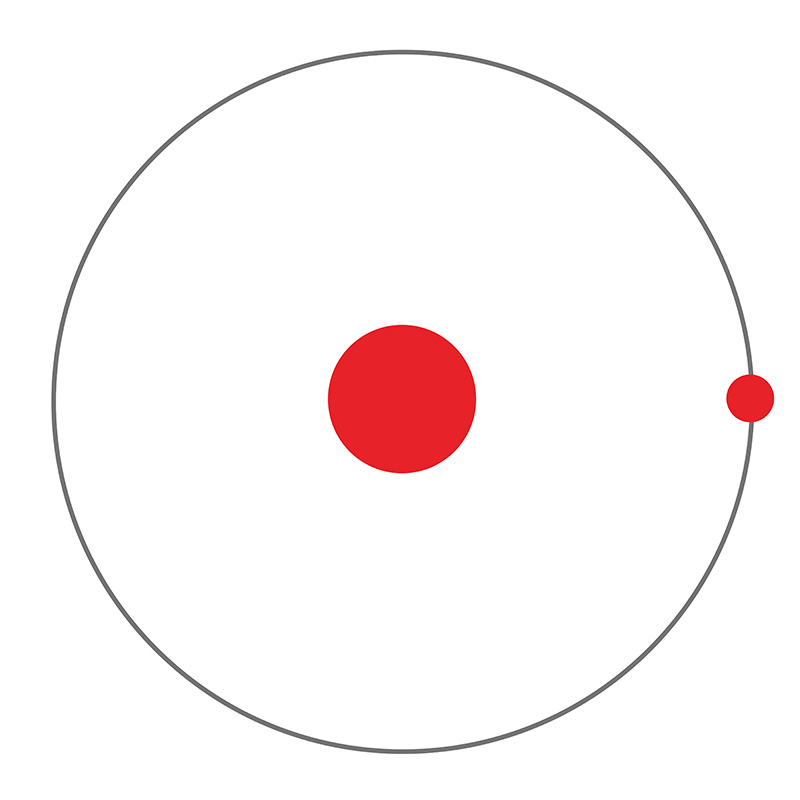
The electrons surround the nucleus and constantly revolve in non-radiating energy orbits known as stationary orbits. The entire atomic mass is concentrated at the centre, where the positively charged particles, called protons, are present. The Bohr Model of hydrogen can be explained by his postulates which are as follows: The formula defines a photon’s energy: E= hf, where h is the Plank’s constant, and f is the frequency. Besides, the Bohr Model formula is defined based on the quantum theory of Planks that says:Įvery matter emits or absorbs a certain amount of energy in small packets known as quantum.įor light energy, a quantum is known as a photon. This is the main reason why his theory is considered as the major game-changer. His theory also proved that the question of instability of the atomic structure as per Rutherford’s model was invalid. In 1913, Niels Bohr introduced the theory about the hydrogen line spectrum and the reasons behind the discontinuities in the lines. On this basis Bohr’s theory of the hydrogen spectrum was conceptualised. The line spectrum is the uniqueness of every element that describes the number of orbits, the energy differences, and electronic behaviour. The emission spectrum represents the energy released by excited atoms when an element is heated. It is a pattern of the atoms’ energy that allows scientists and researchers to study elemental behaviour. It is important to have a basic idea about the atomic spectra to learn everything about the Bohr Model of hydrogen. What is Bohr’s model of the hydrogen atom? Hydrogen atomic spectra This is what led to the conception of the Bohr Model formula. However, when the spectrum was observed in the lab, it was line spectra.Īs Rutherford’s model contradicted the very existence matter, it was considered incomplete. Moreover, it implied that the atomic structure is not stable.Īnother major drawback was his theory about the continuous hydrogen spectra because electrons emit energy constantly. If that had been the truth, then the existence of matter would be under question. 
He said that electrons continuously lose energy and follow a spiral path till it falls into the nucleus. However, his theory had two major drawbacks, which completely contradicted the principle of mass. Rutherford’s gold foil experiment was a huge contribution because he was the first to give an idea about the atomic structure, the positions of the protons and electrons in the atom, and so on. What led to the discovery of Bohr’s Model? Niels Bohr introduced the reality between the atomic structure in the famous Bohr Model formula. However, his theory proved two major flaws- instability of the entire atomic structure and lack of information on the hydrogen spectra. Based on the concentration and deflections of the alpha rays, he concluded that there is a mass of positive charge at the centre of the atom, but surrounding the nucleus are negatively charged electrons. Several years later, Rutherford performed the famous experiment with a radioactive element, radium, and a thin gold foil. Dalton was the first chemist to introduce the fact that matter is made from small elements called atoms, which was considered the smallest form of matter at that time.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
